- Relevant Surgical AnatomyGraham C Calvert, Michael J Beebe, Darrel S Brodke
- Clinical Biomechanics of the Spine: The Nuts and Bolts of Spinal InstrumentationFernando Techy, Edward C Benzel
- Bone and Intervertebral Disk PhysiologyShah-Nawaz M Dodwad, Sohrab S Virk, Safdar N Khan, Tom D Cha, Howard S An
- Preoperative Bone Health Assessment and the Potential use of Systemic Osteoporosis Drugs in Select Patients Undergoing Elective Spine Fusion SurgeryKelly Krohn
- Inflammatory Diseases of the SpineYu-Po Lee
- Principles of Spine Trauma and Spinal Cord InjuryMelissa Nadeau, Brian K Kwon
- Principles of Spinal FusionS Tim Yoon, Colin G Crosby
- Spinal InfectionsScott D Daffner, Vincent J Miele
- Intradural Spinal Tumors
OVERVIEW
The normal human spine is segmented into 7 cervical vertebrae (C1–7), 12 thoracic vertebrae (T1–12), 5 lumbar vertebrae (L1–5), 5 fused sacral vertebrae (S1–5), and 3 or 4 fused coccygeal vertebrae. The spine has four distinct sagittal curves, lordosis (concave posterior curvature) in the cervical and lumbar regions, and kyphosis (concave anterior curvature) in the thoracic and sacral/coccygeal regions. Sagittal balance should be such that, on a standing film, a line drawn vertically downward from the center of the C7 vertebral body intersects the posterior edge of the S1 superior endplate. Sagittal balance is also dependent on the shape of the pelvis and the takeoff of the sacrum (sacral slope) combined with the tilt of the pelvis, which when added together results in the fixed measurement of pelvic incidence. The coronal alignment of the normal human spine is straight. Various congenital, environmental, idiopathic, and degenerative changes can result in deviation from this normal anatomy. Understanding these relationships is paramount when managing spinal deformity.
CORRELATIVE ANATOMY
When performing surgery on the spine and planning incisions, particularly during anterior approaches, it is important to have an understanding of the particular spinal levels that correspond to externally identifiable structures. While these relationships are somewhat variable, they remain an important guide to the underlying spine. Anteriorly, the angle of the mandible overlies the C1–2 level, the hyoid pinpoints the C3 level, the thyroid cartilage overlies the C4–5 level, and the cricoid cartilage and carotid tubercle identifies the C6 level. The vertebral prominens posteriorly corresponds to the C7 vertebra. The T3 level is in line with the manubrium anteriorly and the spine of the scapula posteriorly. The T7 level corresponds to the xiphoid process anteriorly and tip of the scapula posteriorly. The umbilicus overlies the T10 level anteriorly, while the iliac crest overlies the L4 level posteriorly. Other important correlations include the end of the spinal cord and beginning of the conus medullaris at the L1 level, the aortic bifurcation at the L3–4 level, and the bifurcation of the vena cava just below this at the L4 level.
OSTEOLOGY OF THE SPINE
Cervical Osteology
The cervical spine is composed of seven vertebrae, C1 through C7, stacked vertically in a lordotic alignment from the base of the skull to the thoracic spine. The primary function of the cervical spine is to provide support and motion to the cranium while allowing protective passage of the spinal cord, exiting nerve roots, and vertebral arteries. C1 and C2, termed the atlas and axis, respectively, possess unique anatomic characteristics and functions. C3 though C7 form the subaxial region of the cervical spine and are more uniform, possessing classic cervical vertebral anatomy, though C7 may vary slightly with respect as it transitions to the thoracic spine.
The atlas is the most cephalad vertebra forming the junction between the occiput and cervical spine. It uniquely lacks both a vertebral body and spinous process. It is composed of an anterior arch, left and right lateral masses, transverse processes, and a posterior arch. The posterior arch is the first structure encountered during posterior approach and can be absent in up to 5% of the population.1 The posterior ponticulus (aka ponticulus posticus), a boney extension of the posterior arch that surrounds the vertebral artery, may be present in 12–19% of the population.2-4 Preoperative imaging must be examined prior to surgery at this level to rule out these abnormalities and avoid inadvertent breach of both the thecal sac and vertebral artery. The posterior arch contains the posterior tubercle, site of attachment of the ligamentum nuchae, and provides for the wide surface area of the spinal canal at this level with an average sagittal diameter of 23 mm.5 The transverse processes of the atlas have both anterior and posterior tubercles that serve as cervical musculature origins and insertions, respectively. They also contain a foramen transversarium, which serves as a conduit for passage of the ascending vertebral artery. The lateral masses superiorly contain two concave superior facets that articulate proximally with the occipital condyles to form the atlanto-occipital (AO) joint. The inferior surfaces of the lateral masses contain two inferior facets that articulate with the corresponding superior facets of the axis to form the atlantoaxial (AA) joint. The anterior arch contains the anterior tubercle, site of attachment of the anterior longitudinal ligament (ALL), and 4longus colli. The anterior ring possesses a posteriorly facing facet covered with hyaline cartilage, which articulates with the dens of axis.
The axis is the second cervical vertebra and has a more typical vertebral body with disk and facet joint articulation caudally with C3, and a cranial extension, the odontoid process (dens), which along with lateral masses, articulates with C1. The spinous process is the first structure encountered at this level from the posterior approach. It is typically large, bifid, and sometimes palpable through the skin. The laminae at this level are also large and slope laterally and caudally to the lateral masses. The pars interarticularis is formed from the junction of the lateral mass and superior aspect of the lamina. It is confluent with the pedicle of the axis, angling 30° medial and 20° cephalad to attach the posterior boney elements to the vertebral body.6 The pars interarticularis is particularly prone to fracture at this level with a hyperextension load, termed the “hanged man's fracture”. The transverse processes are abbreviated at this level and contain transverse foramina for the traversing vertebral arteries, which take a tortuous path through the pars region, as detailed later in this chapter. The location of the transverse foramen within the processes can be variable and careful examination of preoperative imaging to rule out a medial or “high-riding” vertebral artery is paramount before placing instrumentation (Fig. 1). The lateral masses contain two superior concave and inferior convex facets to allow for rotation with respect to atlas and flexion and extension with respect to C3.
The subaxial cervical spine has typical and uniform vertebral anatomy (Fig. 2). Two distinguishing features of cervical anatomy that differ from more caudal vertebra are transverse foramina and uncinate processes. Transverse foramina in the transverse processes of C3–C6 serve as a conduit for the ascending vertebral artery. The uncinate processes are craniolateral extensions of the superior endplates of the vertebral bodies that articulate with the inferior endplate of the next more superior vertebral body. The anteroinferior edge of the vertebral body typically overhangs the anterior edge of the intervertebral disk at each level and may be removed for improved access to the disk space during anterior discectomy procedures. The lateral masses contain the superior and inferior articular processes and are bordered by the pedicle anteriorly and lamina posteriorly, serving as common instrumentation sites for the posterior cervical spine. The pedicles bridge the posterior boney elements to the vertebral body. The spinous processes of the subaxial cervical spine are small, bifid, and not generally externally palpable. They are directly behind their corresponding vertebrae and project posteriorly and slightly inferiorly.
The seventh cervical vertebra is a transitional segment with slight variations from the above. It has a more prominent and usually palpable spinous process referred to as vertebral prominens. It also has more steeply inclined articular processes similar to thoracic vertebrae. The C7 pedicles are larger in diameter than the other cervical vertebrae and angle posteromedially and inferiorly. The transverse processes of C7 are shorter and typically have rudimentary transverse foramina that generally do not contain the vertebral arteries. Occasionally the anterior projection of the transverse process will develop into an anterior costal projection referred to as a cervical rib.
The thoracic spine is composed of 12 vertebrae, T1 through T12, arranged in a kyphotic alignment that possesses costal facets for diarthrodial articulation with ribs. In general, the thoracic spine is stiffer than the cervical or lumbar spine because of these extra articulations. T2–T8 are considered typical thoracic vertebra and have more uniform characteristics. T1 and T9–T12 are considered transitional vertebra having some features of cervical and lumbar vertebra, respectively. The thoracic spine functions to provide anchoring support to the rib cage, protection to the traversing spinal cord, and transmittal of axial loads.
The vertebral body of a typical thoracic vertebra is kidney-shaped with a larger anteroposterior than medial lateral diameter (Fig. 3). The left side of the vertebral body may take on a flattened appearance due to pulsations from the aorta. The dimensions of the vertebral bodies increase moving down the spinal column. The posterosuperior margins give rise to the thoracic pedicles, while the posterolateral margins of the upper thoracic vertebral bodies contain corresponding superior and inferior costal demifacets for articulation with rib heads. The T11 and T12 bodies, however, possess only one costal facet on each side.
The thoracic pedicles have an oval cross-sectional appearance with the height being greater than the width and the height increases in the more caudal segments. The widths of the pedicles, however, do not follow this pattern, which is important from a fixation standpoint. The T1 and T2 pedicles have relatively large widths along with T11 and T12. The pedicles with the smallest transverse width are found at levels T3–T6. Scoles classically documented the distribution of pedicle widths in a study of 50 cadaveric spines, identifying T6 as having the smallest transverse diameter, with a mean of 3.0 mm in males and 3.5 mm in females. T1 had mean diameter of 6.4 mm in females and 7.3 mm in males. T12 was the largest with a diameter of 7.2 mm in females and 7.4 mm in males.7 The axial plane angulation of the pedicles decreases moving down the spinal column. T1 has a medial angulation of approximately 25°, T2 15°, T3–T10 5–7°, and T11 and T12 have little to no medial angulation. The cortical wall of the medial pedicle is slightly thicker than the lateral wall.
The spinous processes of the thoracic spine arise from the lamina and project posteriorly with caudal angulation. T1 and T2 have longer, more palpable spinous processes than the remaining vertebrae of the thoracic spine. The transverse processes of the thoracic spine possess a concave facet along the anterior margin, which articulates with the tuberculum of the same rib that also articulates with the superior costal facet of the vertebra. The vertebral canal in the thoracic spine takes on a more rounded appearance compared to the typical oval appearance of the cervical and lumbar spine. The diameter of the vertebral canal is also much smaller in thoracic spine measuring only 15–17 mm throughout.8 For this reason, small changes in dimensions of this space from trauma, space occupying lesions, or disk herniations can cause severe spinal cord injuries.
The superior articular facets are oval-shaped structures with a slightly convex surface and project from the junction of the superior lamina and pedicle. They face dorsal and slightly superolateral. The inferior facet manifests from the inferior lamina with a complimentary surface to that of the superior facet, overlapping it in a shingle-like manner. The coronal and slightly oblique orientation of the thoracic facets contribute to resistance to intervertebral shear forces, compressive forces, and intervertebral torsion.9,10 It is also advantageous for lateral bending and accommodates flexion, while limiting rotation.11
Lumbar Osteology
The lumbar spine is normally composed of the five most caudal mobile vertebrae, L1 through L5, occupying the region between the rib bearing vertebrae of the thoracic spine and the fused sacral vertebrae. While spinal segmentation is generally consistent in humans, there is some variation in the transitional regions of T12/L1 and L5/S1. Approximately 12% of the population shows variation in the normal transitional anatomy from L5 to S1, with around 1.8% 6of humans displaying complete lumbarization of the S1 segment (sometimes referred to as L6), while others display sacralization of the L5 segment.12,13 At the cranial aspect, in the region of the thoracolumbar transition, some patients will also display a rudimentary rib arising from the L1 segment.
The normal lumbar spine assumes a lordotic curvature of around 42–45° in the supine position and 50–53° in the standing position beginning at the superior endplate of S1. The convex anterior curvature of the lumbar spine is especially apparent at the lumbosacral junction; where the wedge-shaped L5–S1 disk and L5 body contribute an average of 16° of lordosis.
Characterized by their lack of transverse foramina, costal facets, or intersegmentary fusion, the lumbar vertebrae are the largest and strongest of the dynamic, presacral spine (Fig. 4). Each vertebra consists of a substantial vertebral body with a posteriorly based neural arch for protection of the conus medullaris and cauda equina. The neural arch is formed by a pair of pedicles, a pair of laminae, four articular processes, two transverse processes, and one spinous process. While this is true of a normal vertebra, lumbar vertebrae frequently display any number of congenital or developmental variations due to genetic or environmental factors.
The vertebral body of the lumbar spine is substantially larger than that in the cervical or thoracic spine. Viewed in the axial plane, the body has the shape of a kidney bean, with a convex anterior surface and a concave posterior. When viewed in the axial plane, the width of each body is larger than the depth, with the mediolateral to anteroposterior ratio increasing from around 1.22 at the L1 level to 1.43 at the L5 level.14 The average width of the vertebral body in the coronal plane increases at each level from L1 to L5, while the width of the superior endplate of each segment is less than that of the inferior endplate.14,15 The anteroposterior depth of the vertebral body similarly increases from L1 to L5, with the depth of the inferior and superior endplates of a single segment remaining similar when viewed in the sagittal plane. The lateral and anterior cortices between the inferior and superior endplates are concave in shape, while the posterior cortex gives rise to the neural arch.
The superior and inferior endplates of the vertebral body, as in all nonfused vertebrae, are covered in hyaline cartilage with strong subchondral bone. The edge of each endplate is marked with an area of cartilage free bone, referred to as the ring apophysis, a secondary ossification center of the vertebral body. The ring apophysis serves as the anchoring sight of the Sharpey's fibrils from the disk. The posterior surface of the vertebral body is marked by one or more nutrient foramina, the site of arterial entrance and basivertebral venous exit.
Near the cephalad border of the posterolateral body of each lumbar vertebrae arise a pair of stout pedicles, which form the base of the neural arch. The pedicles contain the strongest bone in the lumbar spine, with the cortical bone of the medial wall being 1.5–2 times thicker than that of the lateral.16 The concave inferior cortex of the pedicle serves as the cephalad border of the intervertebral foramen for the segment's respectively labeled nerve root while the superior cortex of the pedicle serves as the caudal border of the foramen above.
It is crucial to understand the dimensions, orientation, and relationship of the lumbar pedicles to the surrounding osseous features for safe free-hand placement of pedicle screw fixation. In the past, perforation rates have been reported as occurring in 1.8–54.7% of screws placed, with resultant neurologic changes seen in 0–7% of patients.17 Knowledge of the morphologic features will allow the surgeon to select the correct screw starting point, screw length, diameter, and orientation, and thus minimize the occurrence of misplacement.
The lumbar pedicles are generally the widest in the presacral spine. The average width of the L1 pedicle's outer cortex along the short axis (isthmic width) is 8.5 mm increasing to 13.3 mm at L5.18-21 Inner cortical isthmic width likewise increases from 4.6 mm at L1 to 7.9 mm at L5.16 This generally allows for placement of a 6.0 to 7.5 mm pedicle screw in all lumbar pedicles.
The pedicles of the L1 segment have an average medial inclination angle of around 7°.16,21 The posterolateral 7orientation of the pedicles increases to 8°, 13°, 20°, and 32° at L2, L3, L4, and L5, respectively (Fig. 5). The sagittal pedicle inclination angle decreases only slightly from 2.4° to 1.8° from L1–L5, allowing surgeons to use the endplates on a lateral fluoroscopic image for predictable direction of the pedicle screw in craniocaudal plane.21 The pedicle inclination must be considered, particularly in the convergent lower lumbar pedicles, as lack of adequate medialization will commonly lead to a lateral breach. Furthermore, appropriate convergent angulation of the pedicle screw increases pullout strength by more than 28%.22
The depth from the start point of the pedicle to the anterior cortex of the vertebral body (pedicle chord length) averages around 51 mm, a distance varying greatly amongst patients, but only slightly across the lumbar vertebrae of a single subject.16,20,21 While the lumbar pedicle itself only contributes 15–25 mm of this depth, it provides as much as 80% of the caudocephalad stiffness and approximately 60% of the pullout strength.14,16
Many authors have described methods for determining the center of the pedicle in relation to the superficial structures of the neural arch in the lumbar spine.18,23-25 This is vitally important for safe free-hand placement of interpedicular fixation. Authors have recommended starting the screw at a point in line with the articulation of the facet joint at the midpoint of the transverse process,23 at the “nape of the neck,” defined as the lateral and inferior corner of the superior articular facet,25 or at the lateral border of the superior articular process and the midline of the transverse process.24 Recently, Su et al. has recommended surgeons start at a point in line with the most medial aspect of the lateral pars in the horizontal plane and 1 mm superior to the midpoint of the transverse process in the vertical plane.18
Posterior to the pedicle, forming the roof of the neural arch, the lamina of the lumbar spine are short and wide with less overlap than is seen in the thoracic vertebrae. Appreciation of this feature by the surgeon is crucially important to avoid inadvertent penetration into the interlaminar space and resultant dural injury during dissection.
From the superior aspect of each lamina where it adjoins its respective pedicle, arises a superior articular process with a small mammillary process on the posterior border. Emerging from the inferolateral aspect of the lamina is the inferior articular process. In approaching the posterior lumbar spine, it is important for the surgeon to keep in mind the orientation of each vertebra's articular processes to avoid unintended capsular disruption and subsequent destabilization. The superior articular processes exhibit vertical concave articular facets facing posteromedially, while the inferior articular processes exhibit a convex articular surface facing anterolaterally. Together, the ipsilateral inferior and superior articular facets from two adjoining vertebrae form a single facet joint.
The portion of lamina lying between a single vertebra's ipsilateral superior and inferior facet joints and connecting to the pedicle, known as the pars interarticularis, is subjected to considerable bending forces as the energy transmitted by the lamina undergoes a change of direction into the pedicle. To withstand these forces, the cortical bone of the pars interarticularis is generally thicker than other laminar bone in the lumbar spine.26,27 However, in 4–6% of individuals, despite this cortical thickening, the bone is insufficient to withstand the transmitted forces resulting in fatigue and stress fractures of the pars interarticularis.28-30
Arising from the posteromedial meeting point of a lumbar vertebra's laminae is a quadrangular spinous process. In the lumbar spine, the spinous process is aimed almost directly dorsal with little caudal slope and it is thickened along its posterior and inferior borders.31 The spinous processes of the cranial four lumbar vertebrae are similar, with the fifth being the smallest, baring a rounded and down-turned apex.
8Protruding laterally from the junction of a pedicle and its respective lamina is a flat, rectangular transverse process. The transverse processes of the first three lumbar segments are thin and long, projecting laterally and slightly posterior, increasing in length at each level. Conversely, the fourth lumbar transverse process decreases in length when compared to its cranial brethren. The fifth differs significantly from the superior four transverse processes, in that it passes laterally and then superolaterally to a blunt tip and the whole process presents a greater dorsal inclination than the others.31 Near its attachment to the pedicle, each transverse process bears on its posterior surface a small, irregular bony prominence known as Luschka's accessory tubercle, or the accessory process.27,32 The accessory processes vary in form and size from a simple bump on the back of the transverse process to a more definitive pointed projection of capricious length.27 The accessory process is identifiable as the only bony projection from the posteroinferior root of each transverse process and lays slightly inferolateral to the mammillary process, separated by a space known as the mamilla-accessory notch. Between the accessory and the mammillary process, on the dorsolateral facet, runs a ligament known as the mammilloaccessory ligament, which forms the roof of a tunnel through which the medial branches of the posterior primary ramus course.
As in the cervical and thoracic spine, the lumbar spinal canal is bounded dorsally by the lamina and ligamentum flavum, ventrally by the vertebral body, intervertebral disk, and posterior longitudinal ligament (PLL), and laterally by the pedicles and intervening foramina. The shape of the intervertebral canal of the lumbar spine is ellipsoid proximally and gradually becomes triangular at the caudal levels. While the majority of the canal is filled with the thecal sac, the epidural space is principally filled by a thin layer of connective tissue, sometimes referred to as the epidural membrane. The normal lumbar spinal canal has a mid-sagittal height of around 18–20 mm and a cross-sectional area of around 200–225 mm2, a value that increases by approximately 24 mm2 in flexion and decreases by 26 mm2 in extension.33,34 Symptomatic stenosis has been proposed by Schonstrom et al. to occur at areas of less than 100 mm2 or mid-sagittal height less than 10 mm.35
The radicular canal is the lateral gutter of the spinal canal containing the nerve root, from its emergence through the thecal sac to its exit out the intervertebral foramen.36 It is formed by a bony concavity, initially directed toward the midline then inferior, that houses the nerve as it exits the spinal canal. The radicular canal can be divided into three parts: retrodiscal, parapedicular, and foraminal. The retrodiscal segment refers to the zone wherein the traversing nerve crosses the disk above its foraminal exit. The second section, the parapedicular segment, commonly known as the lateral recess, runs the entire height of the medial pedicle. Finally, the foraminal portion of the radicular canal is formed by the inferior pedicle as the nerve exits the intervertebral foramen.
In cadaveric studies of lumbar foramina, reported foraminal dimensions vary significantly.34,37,38 Foraminal height averages around 18–20 mm, while sagittal depth averaged 8–9 mm superiorly and 3–4 mm inferiorly. Together these parameters contributed to an osseous cross-sectional area of around 70–130 mm2, which is significantly larger than that of the nonosseous area, even in the normal spine.38 While Hasegawa reported that significant stenosis was noted in 4 of 5 specimens with a foraminal height below 15 mm and 8 of 10 with a posterior disk height below 4 mm,37 only the latter holds true in a similar study by Inufusa et al.34 They reported significant stenosis in those cadavers with decreased posterior disk height, midforaminal width, and foraminal cross-sectional area.
Sacral and Coccygeal Osteology
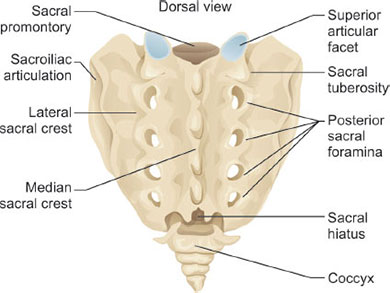
Triangular in nature and formed by five fused vertebrae, the sacrum functions to transmit loads from the dynamic presacral spine to the pelvic girdle and subsequently the lower extremities. While the sacrum is generally a single fused structure, it is important for the surgeon to continue to appreciate the unique markers of each sacral segment to avoid inadvertent nerve injury.
Anteriorly, a longitudinal protuberance represents the fused sacral bodies, the superior most portion of which is known as the sacral promontory. Along this protuberance, transverse apices represent the ossified or vestigial disks. Terminal fibers of the anterior and posterior longitudinal ligaments are attached to the ventral and dorsal surfaces of the first sacral body.31 Posteriorly a series of midline prominences represent the successively smaller spinous processes. The laminae of each level fuse medially, while laterally the transverse processes fuse to form the lateral mass. Between the fused laminae and transverse processes a set of anterior and posterior foramina arise, which transmit the ventral and dorsal rami, respectively (Fig. 6).
Near the inferomedial edge of each posterior foramen, a small articular tubercle is noted, representing the fused zygapophyseal joint.27 The consecutive series of articular tubercles constitutes the intermediate crest of the sacrum. Far laterally, the lateral crest of the sacrum is formed by a longitudinal series of transverse tubercles, representing the tip of each fused transverse process.
The superior border of the sacrum is of particular interest to the spine surgeon as many constructs extend to the S1 segment. The superior sacral surface presents 9a set of posterior facing and coronally oriented articular surfaces, the intact superior articular processes of the S1 segment. In conjunction with the inferior processes of the L5 vertebrae, these form the L5–S1, or lumbosacral articulation. Superolaterally, the transverse processes of the S1 segment extend further lateral than those of the subsequent sacral segments to form the sacral ala.
The laminae of the fifth sacral segments fail to fuse at the midline, forming the sacral hiatus. At the lateral edges of the sacral hiatus, the inferior articular processes of the S5 segment form the cornua, which articulate with the three to four fused coccygeal segments.
On the ventral surface, the lateral aspect of the ala forms an attachment point for the medial aspect of the iliacus. The lateral mass of the second through fourth segments serves as the origin of the piriformis. The lateral edge of the fifth segment supports the origin of the coccygeus.
On the dorsal surface, the lateral aspects of the fourth and fifth segments serve as an attachment point for the medial aspect of gluteus maximus. Medial to and between the first through fourth dorsal foramina, the multifidus seats its sacral origin. Around the edge of the multifidus origin, the erector spinae aponeurosis attaches.
The sacrum plays a critical role in terms of the overall sagittal alignment of the spine. It is important for the surgeon to have a good understanding of both normal and pathologic alignment of the sacrum in order to assess and treat global sagittal deformity. Three important parameters need to be assessed for complete understanding of sacral alignment: sacral slope, pelvic tilt, and pelvic incidence (Fig. 7). Sacral slope is the angle between the S1 superior endplate and the horizon. Pelvic tilt is the angle between a vertical line and a line connecting the center axis of the femoral heads and the center of the S1 superior endplate. These two parameters are variable and have an inverse relationship with one another. Pelvic incidence is the angle formed between a line perpendicular to the S1 superior endplate and a line between the center axis of the femoral heads and the center of the S1 superior endplate. Pelvic incidence is a morphologically fixed parameter and can be calculated as the sum of the sacral slope and pelvic tilt. Normal pelvic incidence in the asymptomatic adult population is 55° ± 10.6°. The correlation between pelvic incidence and lumbar lordosis has been well described.39 In general, those patients with high pelvic incidence typically have a vertical sacrum and a high lumbar lordosis. Conversely, those with low pelvic incidence typically have a more flat sacrum and less lumbar lordosis. Consideration of these relationships is the key to assess global sagittal balance prior to any deformity procedure.
While the coccyx provides no structural support, it serves as an attachment for multiple muscles. The gluteus maximus attaches to the dorsal surface, while the levator ani and sphincter ani attach to the tip, and the coccygei attach to the lateral edges.
ARTHROLOGY OF THE SPINE
Special Articulations of the Cervical Spine
The AO articulation is responsible for allowing 25° or up to 50% of the total flexion and extension motion of the cervical spine.6 The rounded occipital condyles form a ball and socket configuration with the cup-shaped superior articular surface of atlas. The lateral aspect of the articular surface of the atlas is steep to provide containment of the occipital condyles and prevent lateral displacement.
The AA articulation is composed of two lateral joints and one median joint and is responsible for up to 50% of rotatory 10motion of the cervical spine. The median joint is referred to as a double joint: the anterior odontoid articulation with the posterior surface of the anterior atlas arch and the anterior surface of the transverse ligament articulation with the posterior surface of the odontoid. The transverse ligament of the atlas is the principal stabilizer of the AA joint. It spans the posterior arch of the atlas and possesses superior and inferior extensions referred to as the cruciform ligament. The transverse ligament is on average 21.9 mm in length, 6–7 mm in height, and able to withstand a 350 N load.40-42 A fibrocartilaginous surface coats the anterior surface of the ligament to facilitate a dynamic pivot interaction with the odontoid. Traumatic rupture of this ligament can lead to AA instability and migration of the dens toward the brain stem. The ligament is prone to congenital laxity in diseases such as Down's syndrome and chronic laxity in diseases such as rheumatoid arthritis.
The secondary stabilizers of the AA articulation are the alar ligaments and tectorial membrane. The alar ligaments are a pair of ligaments that attach the superior aspect of the odontoid to the skull base. They not only stabilize but also limit rotation and lateral bending to the contralateral side of the AA articulation. These ligaments become the primary stabilizers of the AA articulation should the transverse ligament rupture, and have the ability to withstand a 200 N load.42
The tectorial membrane is a broad, layered ligament that lies dorsal to the cruciform ligament attaching to the skull base cranially and continuous with the posterior longitudinal ligament caudally. It too is a secondary stabilizer to the AA joint able to withstand a 76 N load before failure.43 The facet joints of the AA joint are made up of the inferior, caudally projecting medial to lateral articular processes of atlas and the corresponding superior, cephalad projecting lateral to medial articular processes of axis. The surfaces are rounded with respect to each other to allow rotatory motion between them (Fig. 8). The capsules of these joints also act as stabilizers of AA motion.
Intervertebral Joint and Disk
At the junction of any two vertebrae, three joints control movement: the intervertebral joint (the disk /endplate complex), and the bilateral zygapophyseal or facet joints. The first, and most sizeable, is the intervertebral joint between the two vertebral bodies. The intervertebral joint is decidedly specialized, not only permitting multidirectional motion between vertebral segments, but also absorbing and transmitting sizeable, repetitive loads. A single intervertebral joint consists of two vertebral endplates and a single intervertebral disk. Between the two endplates, the intervertebral disk consists of two major components, the nucleus pulposus and the annulus fibrosus, each highly differentiated in both form and function. The posteriorly-eccentric, central component of an intervertebral disk is the proteoglycan-rich nucleus pulposus, constituting the central 50–60% of the diameter, while the peripheral component, the collagen-rich annulus fibrosus, comprises the remaining 40–50%.44 The major constituent of the in vivo intervertebral disk is water, making up 66–86% of the volume, with maximal concentration in the nucleus and decreasing concentration toward the periphery of the annulus.44,45 Most of the remaining disk is composed of extracellular matrix, primarily collagen and proteoglycans. Fibroblasts and chondrocytes make up approximately 1% of the disk.46 Nerves and vessels, while present in the disk, only extend into the outer few millimeters of the annulus.47
The annulus fibrosis, the outer band of tissue of the intervertebral disk, offers high tensile strength due to its highly organized structure. It is made up of a series of 15–25 concentric lamellar rings.48 Each lamella is 0.1 to 0.5 mm thick and composed of parallel collagen fibrils arranged 60° off the vertical axis and alternating left to right in a crisscross pattern between adjacent lamellar rings. The majority of the outer annulus is composed of type I collagen with some of the collagen fibers inserting into the endplate, while others merge with the periosteum and fibers of the ALL and PLL. In the inner annulus, the transitional zone, there is a shift to type II collagen before reaching the nucleus.49 These transitional collagen fibrils of the inner annulus insert into the mineralized endplate.50 Functioning in conjunction with the collagen fibrils of the lamellae to absorb radial loads, elastin fibrils make up around 2% of the dry weight of the annulus, with increasing density in the outer margin.51 Elastin fibrils not only run in parallel with the collagen fibrils of the lamellae, but also connect the adjacent lamellae helping 11to prevent delamination with torsional loads.52 Finally, the annulus contains a large assortment of various other structural glycoproteins, such as fibronectin, laminins, and tenascins.53
The nucleus pulposus forms the less organized, amorphous-appearing center of the intervertebral disk. Unlike the cells of the annulus, which produce both type I and II collagen in a systematic pattern, the cells of the nucleus produce only type II collagen in an irregular pattern, which serve to link the proteoglycan aggregates.49 Proteoglycan aggregates are formed from glycosaminoglycan (GAG) chains of chondroitin sulfate and keratan sulfate covalently bonded to a polypeptide core protein.54 The negatively charged GAG side chains attract cations, which in turn contribute to a high level of water retention.53 These negatively charged chains also interact with other matrix molecules and soluble factors. The irregular pattern of collagen and proteoglycans within the nucleus pulposus allows for absorption of large axial loads through deformation. Much of this force's vector is converted from an axial direction to a radial direction and dispersed into the annulus. The radial dissipation of the energy by the intervertebral disk results in a load of 60–80 kg/cm2 across the posterior annulus, putting in place optimal conditions for the commonly occurring tears of the posterior annulus and subsequent herniation of the pulposus.55
The vertebral endplate consists of cortical bone covered by a thin layer of hyaline cartilage, on average around 0.6 mm thick and thinner toward the center.56 The endplate is essential to disk health as the majority of the disk, other than the outer edge of the annulus, receives nutrients from diffusion through the endplate.47,57 Obstruction of this diffusion, as seen in calcification of the chondral layer in the older population, has been linked to relative nutrient deficiency in the disk resulting in degeneration.57 The endplate is also vital to joint stability in both traction and rotation. The ring apophysis, at the periphery of the endplate, is the most stable lamellar insertion from the annulus into the vertebra. In the cervical spine, the inferior endplates are concave in the coronal plane while the corresponding superior endplates are convex. In the thoracic and lumbar spine, the superior and inferior endplates are both slightly concave. This is important to understand when performing discectomy work in preparation for fusion or replacement.
The interbody joints of the lumbar spine have a higher axial area than those of the prelumbar spine, an essential characteristic to absorb the large loads transmitted across the lumbar intervertebral disks.27 Nachemson et al., in a series of studies, showed that the lumbar disks are subjected to loads of around three times the patient's upper body weight in the sitting position.55,58 These loads were about 30% less in the standing position, about 50% less in the reclining position, and about 30% greater in the forward leaning position. Miller et al. showed that these loads result in histologically notable disk degeneration first appearing in the second decade of life in males and third decade in females.59 By the age of 50, more than 97% of all lumbar disks are degenerated, even in asymptomatic patients.
With aging, the boundary between the annulus fibrosis and nucleus pulposus becomes less distinct. The nucleus pulposus gradually loses its proteoglycan content, becomes fibrotic, and loses its water retaining properties.60 Likewise, the collagen within the lamella of the annulus fibrosis becomes less organized resulting in irregular interdigitation of collagen and elastin networks making them prone to cleft formation and fissuring. These processes account for changes in loading characteristics of the disk with a tendency to lose height and bulge under axial loads.61 They can progress to pathologic conditions such as radial tears in the annulus fibrosis with herniation of the nucleus pulposus or chronic disk bulging with encroachment on the spinal canal.
Zygapophyseal Joints
The zygapophyseal joints (facet joints) of the subaxial spine are diarthrodial joints made up of cephalad projecting, anterior facing inferior articular processes articulating with superior projecting, posterior facing superior articular processes with a surrounding capsule and intervening disk. Anatomic studies demonstrate an average facet joint width of 11 mm with a cartilage thickness ranging from 0.4 mm to 0.9 mm with females having thinner measurements.62,63 This difference has been postulated to have clinical effects in traumatic situations. The facet joints of the cervical spine are typically angulated 45° in the sagittal plane from the transverse process with a tendency to move toward the horizontal at more caudal levels. The facet capsules are robust and play a large role in cervical stability with the ability to withstand a 61 N load before failure.64 White and Panjabi classically demonstrated that sectioning the posterior facet capsules produced instability with translation of the vertebral bodies greater than 3.5 mm under normal physiologic loads.65
Similar to those of the cervical spine, the facet joints of the thoracolumbar spine are true synovial joints with hyaline cartilage on the articular surfaces and a joint space enclosed by a fibrous capsule. On the superior and medial aspects, the facet capsule blends with the ligamentum flavum. Along the dorsal aspect of the joint, the capsule is about 1 mm thick with the outermost fibers of the capsule attaching about 2 mm from the edge of the articular cartilage.66,67 This dorsal capsule is also reinforced by some of the deep fibers of the multifidus muscle.27 Both superiorly and inferiorly, the capsule is more capacious creating subcapsular pockets, which in the intact joint are filled with fat.68
12Functionally, the facet joints of the lumbar spine have been found to carry 3–35% of the static compressive load and up to 33% of the dynamic load.69-72 The orientation of the facet, along with the concavity of the superior articular process and convexity of the inferior surface, also plays an important role in allowing flexion and extension, but limits rotation and forward translation of the superior vertebrae on its inferior mate. The inferior facets of a lumbar vertebra lock themselves between the superior facets of the next caudal vertebra. Generally, the facet joints of the lumbar spine are oblique when viewed through an axial cut, with the shape of the joint assuming a “C” or “J” shape with the concavity pointing posteromedial. The angle of the facet joints become increasingly less sagittal and more coronal from L1–2 to L5–S1, with angles off the sagittal plane averaging 25°, 28°, 37°, 48°, and 53° for L1–2 through L5-S1, respectively.73-75 When the joint takes on a more sagittal orientation it may diminish the protection it offers against forward translation, particularly following surgical decompression, becoming a risk factor for the development of spondylolisthesis and isolated facet arthritis.76-79 Likewise, greater interfacet width, shorter interfacet height, and shorter, narrower articular facets, have all been shown to be independent risk factors for development of spondylolysis.80
Spinal Ligaments
Each functional spinal unit is connected by seven unique ligaments from C2 to the sacrum: the anterior longitudinal ligament, the posterior longitudinal ligament, the ligamentum flavum, the interspinous ligament, the bilateral intertransverse ligaments, the ligamentum nuchae that gives rise to the supraspinous ligament, and the bilateral capsular ligaments (Fig. 9).81 The spinal ligaments are primarily collagenous except for the ligamentum flavum, which is comprised mainly of elastin.27
The ALL originates at the base of the occiput and extends the length of the spine along the anterior aspect. In dissection, the ALL is wider at the level of the vertebral body and narrower at the disk level. Fibers of the ALL incorporate into the periosteum and anterior vertebral bodies of each vertebra, as well as to the annulus of each intervertebral disk.82 The deep fibers of the ALL connect only a single level, while those of the superficial level may connect 3–5 levels. Functionally, the ALL is a strong, thick ligament that resists hyperextension with a load to failure of around 590 N.83
The PLL extends the length of the spine along the posterior aspect of each vertebral body, anterior to the spinal cord. Opposite the ALL, the PLL has a wider attachment at the disk level and narrower attachment at the level of the body.81 The PLL is less adherent to the posterior body than the ALL is to the anterior and bowstrings across the vertically concave posterior body. At the level of the disk, the PLL is intimately associated with the posterior medial fibers of the annulus but this attachment thins laterally toward the uncinate process resulting in the most common site for a cervical disk herniation. The PLL is prone to abnormal calcification in the cervical spine resulting in a dysfunction termed ossification of the posterior longitudinal ligament (OPLL). Tsuyama has reported the incidence of OPLL as 2.4% in Asian populations with 0.16% in non-Asian populations.84 The disease process is a known cause of cervical myelopathy and controversy exists over the approach to the cervical spine when this disease process is present.
The ligamentum flavum (LF), Latin for “yellow ligament”, lies immediately posterior to the thecal sac. It originates bilaterally on the anteroinferior aspect of the lamina of the superior vertebra and inserts on the posterosuperior aspect of the lamina of the inferior vertebra. It is often difficult to appreciate the bilateral nature of the ligamentum flavum during surgical dissection, as it appears as one ligament across midline. It is composed largely of elastic fibers that decrease with age, resulting in hypertrophy and occasionally infolding toward the spinal cord leading to stenosis of the vertebral canal. This ligament has been shown to have a load to failure of 353 N in the cervical spine.83
The interspinous ligament (IL) is a thin, weak membranous ligament that lies interposed between the spinous processes. It functions in resistance of hyperflexion and is easily torn in traumatic situations. The bilateral intertransverse ligaments are likewise interposed between the transverse processes and are usually encountered and preserved during dissection for a posterolateral fusion.
The ligamentum nuchae exists in the cervical spine and lies posterior to the spinous processes and confluent with 13the supraspinous ligament. It possesses two layers, a dorsal raphe, and a ventral midline septum. The dorsal raphe is a thick strip of collagenous tissue formed by intervening fibers of the upper trapezius, splenius capitis, and rhomboid minor. It is firmly attached to the external occipital protuberance cephalad and the C7 spinous process caudad. This portion has a small attachment to the C6 spinous process but no attachments to the more cephalad spinous processes. The ventral midline septum consists of a thin strip of connective tissue that extends anterior from the dorsal raphe and attaches to the more proximal C2–C6 spinous processes and confluent with the interspinous ligaments and AA, AO membranes.85
The supraspinous ligament originates from the ligamentum nuchae at C7. It is the bipedal equivalent of the paxwax ligament in quadrupeds, and extends the length of the spine connecting the posterior apices of the spinous processes. Finally, the capsular ligaments surround each facet joint. The SL, IL, and capsular ligaments are part of the posterior ligamentous complex, which plays an important role in spinal stability with respect to trauma.
NEURAL ELEMENT ANATOMY AND RELATIONS
The spinal cord runs in the vertebral canal throughout the spine giving 31 pairs of nerve roots throughout its course. In total, there are 8 pairs of cervical nerve roots, 12 thoracic, 5 lumbar, 5 sacral, and 1 coccygeal pair. The nerve roots exit the vertebral canal through the intervertebral neuroforamina. The neuroforamen is shaped like a funnel with a narrow medial entrance and wider lateral exit. The nerve root is made up of dorsal and ventral roots within the dural sheath and comprises approximately one quarter to one-third of the cross-sectional diameter of the neuroforamen.86 In the cervical spine the nerve roots exit the vertebral canal above the corresponding pedicle or ring for which they are named. For example, the C1 nerve roots exits above the C1 ring, the C2 nerve roots exits above the C2 pedicle, and so on. The C8 nerve root is the exception, exiting above the T1 pedicle, as there are only seven cervical vertebrae. The remaining roots of the mobile thoracic and lumbar spine exit below the pedicle for which they are named. For example, the T1 nerve root exits below the T1 pedicle, the T2 exits below the T2 pedicle, and so on to L5.
While the conus medullaris, the caudal aspect of the intact cord, may terminate anywhere between T12 and L3, it usually ends between the first and second lumbar vertebrae, with average being at the level of the middle third of L1.87 The conus medullaris of females, African-Americans, and elderly tends to terminate, on average, slightly more distal than those of young, white males.88
Distal to the conus, the rootlets of the cauda equina do not lie randomly, but are held in position by invaginations of arachnoid.89,90 The rootlets in the thecal sac lie in a reproducible, layered pattern, analogous to the laminar configuration of the nerve fibers in the tracts of the spinal cord, with the sacral rootlets at the dorsomedial surface and those contributing to the next exiting root most lateral. Each nerve root is formed by 2 to 12 nerve rootlets within the thecal sac.27,91 The nerve roots from L1 to L5 emerge from the lateral aspect of the thecal sac sequentially, within a dural sleeve, at an inferolateral angle of about 40° from vertical. At the first sacral level, there is an abrupt increase in the acuity of the takeoff to around 22°, with each lower sacral root increasing acuity even further. This is thought to be due to the ascensus medullae spinalis, the cranial migration of the lower cord due to increased growth of the osseous spine in comparison to the cord during development.92
As the dorsal and ventral roots of a given level exit the thecal sac, they closely hug the inferomedial surface of the pedicle. The dorsal root ganglia can be found directly inferior to the pedicle as the nerve passes through the foramen, sometimes even overlying the lateral aspect of the disk.93 At the lateral aspect of the pedicle, the roots converge to form a spinal nerve and then immediately diverge into a larger ventral ramus and a smaller dorsal ramus as they exit the foramen.
Within the vertebral canal, the thecal sac and the nerve root sleeves are secured to the vertebrae by thickenings of the epidural fascia known as dural ligaments or meningovertebral ligaments.27 These ligaments hold the thecal sac to the ventral surface of the canal until its termination, the filum terminale, which generally occurs at the upper third of the S2 body; although the level of termination can range from the lower third of L3 to the upper third of S5.87
The pelvic sympathetic trunk is formed by four of five interconnected sacral ganglia. Superiorly, it is connected to the lumbar sympathetic trunk and inferiorly it terminates in the ganglion impar anterior to the coccyx, which can be treated with an injection or nerve block to potentially relieve coccydynia.94 Branches from the first two sacral ganglia form the inferior hypogastric plexus, which connects to the superior hypogastric plexus through the hypogastric nerve. Several other branches form a plexus on the median sacral artery.
VASCULAR ANATOMY
Cervical Spine
The vertebral artery is the major blood supply to the brainstem and cerebellum, contributing to the Circle of Willis and providing for the blood supply to the remainder of the 14brain should the carotids become compromised. It is divided into four sections: V1–V4.95 The V1 portion spans the take-off point from the subclavian artery to the C6 transverse foramen. The V2 portion spans the C6 transverse foramen to the C1 transverse foramen. The V3 portion spans the exit point of the C1 transverse foramen to the entry point into the foramen magnum. The V4 portion is the intracranial portion where the artery pierces the dura, joins the contralateral artery, and forms the basilar artery. When performing anterior cervical procedures, it is important to have a good understanding of the V2 portion of the vertebral artery. Throughout this section, the vertebral artery lies lateral to the uncinate process just anterior to the nerve root at the middle third level of the vertebral body. An anatomic study by Russo demonstrated an average 1.3 mm gap between the vertebral artery and uncinate process from C3–C6.96 From C6 to C3 the vertebral artery ascends medially with an angle of 4° referenced to midline and it lies slightly more posterior at C3 than at C6.97 Therefore, one should exercise caution with anterolateral decompression at more caudal levels. At the C6–7 level, the vertebral artery lies between the transverse process and longus coli before entering the C6 transverse foramen. At this level, extensive lateral dissection of the longus coli could risk injury. In summation, when performing anterior surgery it is paramount to examine preoperative imaging to identify the entry point and course of the vertebral arteries as they are variable. It is also crucial to identify the uncinate process when doing disk preparation work and stay within the confines of this structure.
At the AA level, the artery takes a series of tortuous turns before entering the foramen magnum. In 80% of the population, the vertebral artery takes an acute lateral bend in the lateral mass of C2 under the superior articular process.98 This bend can occur very high and medial within the lateral mass in some individuals, termed a “high-riding vertebral artery”, and is prone to injury during C2 pedicle screw, pars screw, or C1–2 transarticular facet screw placement. Studies have shown that 18–20% of the population has a “high-riding vertebral artery”.99,100 Just after emerging from the C1 foramen transversarium, the vertebral artery takes another turn 90° medial and runs horizontal within the groove of C1 toward the medial edge of the posterior ring before once again turning cephalad toward the foramen magnum. In this area, the artery is at particular risk of injury during exposures for decompressive or fusion procedures. Ebraheim suggested that one should limit lateral dissection of the posterior ring of C1 to 12 mm posterior along the ring and 8 mm along the superior aspect of the ring to avoid vertebral artery injury.101 A bridge of bone termed the Ponticulus Posticus (Latin for “little posterior bridge”) can envelope the vertebral artery as it traverses the groove at C1. This anomaly exists in about 15% of the population and is important to recognize on preoperative imaging as it can be mistaken as a widened posterior arch during exposure, resulting in injury to the vertebral artery during C1 lateral mass instrumentation.3
When performing cervical spine procedures, it is also important to understand the anatomy of the carotid artery, the major blood supply to the head and neck. They run vertically in the neck and within the carotid sheath, which also contains the internal jugular vein and vagus nerve. The artery bifurcates into the external carotid artery and internal carotid artery (ICA) around the C3 or C4 level. During an anteromedial approach to the cervical spine pulsations of the carotid artery within the sheath are palpable, helping to identify this structure so it can be retracted laterally to avoid injury. The ICA is particularly prone to injury during C1, C2 transarticular, and C1 lateral mass fixation because of its proximity to the anterior aspect of these levels. In an anatomical study, Currier et al. defined the closest distance between C1 and the internal carotid artery as 2.8 mm.102
Blood Supply to the Thoracic Spine
The thoracic aorta runs on the left side of the thoracic spine in an anterolateral position, spanning the T4–T12 segments in straight spines. Kuklo et al. documented a slight movement of the descending aorta toward midline at the more caudal levels of T11 and T12 with a 2.8 mm average distance from the T11 body.103 They went on to demonstrate that in spinal deformity the descending aorta takes on a more posterolateral position to the left of the vertebral body at the apex of the curve (right thoracic curves), but quickly moves toward midline at the T11 and T12 levels perhaps creating a tethering effect as it traverses the aortic hiatus of the diaphragm. The position of the thoracic aorta is important to understand in both straight and spinal deformity patients when performing instrumentation at these levels.
The thoracic aorta gives off paired intercostal arteries at each level with the right intercostal artery being longer than the left given the left-sided position of the aorta. The intercostal arteries further branch into a posterior and spinal branch before coursing around the thoracic spine and lying posterior to each rib. The posterior branch further divides into the internal branch, which supplies the paraspinal muscles, and the external branch, which supplies the skin. The spinal branch enters the vertebral foramina and branches into anterior and posterior radicular arteries that run with the ventral and dorsal rootlets, respectively. The anterior radicular arteries persist and coalesce to form the anterior segmental medullary artery that anastomoses with the anterior spinal artery. The anterior spinal artery runs vertically in the central sulcus of the spinal cord, providing for the anterior two-thirds of its blood supply. The posterior radicular arteries feed the right and left posterior spinal 15arteries, which also run vertically along the posterolateral aspect of the spinal cord providing the posterior one-third of its blood supply.
The artery of Adamkiewicz is the largest of the anterior segmental medullary arteries and provides the greatest contribution to the anterior spinal artery. It arises most commonly on the left side at the T7 to T12 level with T10 being the most common.104 Although it is only one of many segmental arteries, due to its size, compromise of this vessel alone can result in anterior spinal cord syndrome.
Blood Supply to the Lumbar Spine
Discussed earlier in this chapter, the blood supply of L1 through L4 is typical, in that each segment receives its blood supply from a pair of lumbar segmental arteries directly branching from the aorta. However, the blood supply to L5 and the sacrum, from the iliolumbar artery and the median and lateral sacral arteries, is unique and deserves special attention.
The iliolumbar artery arises as the first branch of the dorsal division of the internal iliac artery. Its path begins as it heads superiorly dorsal to the obturator nerve and ventral to the lumbosacral trunk, the connection between the lumbar and sacral plexuses.105 As it reaches the medial border of the psoas at the inferior margin of the L5–S1 disk, it divides into an iliac and a lumbar branch.
The iliac branch descends to supply the iliacus muscle. Its anastomosis with the obturator artery supplies the bone of the ilium, while also giving off distributing branches to the gluteal and abdominal muscles. It then further anastomoses with the superior gluteal artery, iliac circumflex artery, and the lateral circumflex femoral artery.
The lumbar branch continues its ascent on the posterolateral surface of the L5–S1 disk, supplying the psoas and quadratus lumborum along its path.106 Cranially, the lumbar branch anastomoses with the L4 segmental artery and in conjunction they supply the spinal vessel to the L5–S1 foramen.
The median sacral artery arises from the dorsal aorta proximal to its bifurcation. It descends in the midline in front of the L4 and L5 vertebral bodies giving off branches, which anastomose with the lumbar branch of the iliolumbar artery as well as the lateral sacral arteries.
The lateral sacral arteries are a bilateral set of superior and inferior arteries. In 93% of specimens, the lateral sacral arteries arise as the second dorsal division of the inferior iliac arteries, with the other 7% arising directly from the internal iliac.107 In 51% of specimens, the arteries arise as a common trunk then further dividing into superior and inferior branches, while in 47% of specimens they represent two completely distinct arteries.108 The superior lateral sacral artery anastomoses with branches from the median sacral artery and together they enter the first or second anterior sacral foramen. The inferior branch travels across the front of the piriformis and sacral nerves to the medial side of the anterior sacral foramina where it then descends supplying branches to the third and fourth anterior sacral foramina. The branches from both the superior and inferior lateral sacral arteries, which enter the anterior foramina, supply nutrients to the contents of the sacral canal, then exit through the posterior sacral foramen to supply the muscles on the dorsum of the sacrum and finally anastomose with the superior and inferior gluteal arteries. In their path, the lateral sacral arteries also supply branches to the piriformis and coccygeus muscles, the sacral plexus, and the rectum.
REFERENCES
- Gehweiler JA, Daffner RH, Roberts L. Malformations of the atlas vertebra simulating the Jefferson fracture. AJR Am J Roentgenol. 1983;140(6):1083–6.
- Hong JT, Lee SW, Son BC, et al. Analysis of anatomical variations of bone and vascular structures around the posterior atlantal arch using three-dimensional computed tomography angiography. J Neurosurg Spine. 2008;8(3):230–6.
- Young JP, Young PH, Ackermann MJ, et al. The ponticulus posticus: implications for screw insertion into the first cervical lateral mass. J Bone Joint Surg Am. 2005;87(11):2495–8.
- Hasan M, Shukla S, Siddiqui MS, et al. Posterolateral tunnels and ponticuli in human atlas vertebrae. J Anat. 2001;199(Pt 3):339–43.
- Ebraheim NA, Lu J, Yang H. The effect of translation of the C1-C2 on the spinal canal. Clin Orthop Relat Res. 1998;351:222–9.
- Xu R, Nadaud MC, Ebraheim NA, et al. Morphology of the second cervical vertebra and the posterior projection of the C2 pedicle axis. Spine (Phila Pa 1976). 1995;20(3):259–63.
- Scoles PV, Linton AE, Latimer B, et al. Vertebral body and posterior element morphology: the normal spine in middle life. Spine (Phila Pa 1976). 1988;13(10):1082–6.
- Masharawi Y, Salame K. Shape variation of the neural arch in the thoracic and lumbar spine: characterization and relationship with the vertebral body shape. Clin Anat. 2011;24(7):858–67.
- Adams MA, Hutton WC. The effect of posture on the role of the apophysial joints in resisting intervertebral compressive forces. J Bone Joint Surg Br. 1980;62(3):358–62.
- Adams MA, Hutton WC. The mechanical function of the lumbar apophyseal joints. Spine (Phila Pa 1976). 1983;8(3):327–30.
- Masharawi Y, Rothschild B, Dar G, et al. Facet orientation in the thoracolumbar spine: three-dimensional anatomic and biomechanical analysis. Spine (Phila Pa 1976). 2004;29(16):1755–63.
- Bron JL, van Royen BJ, Wuisman PI. The clinical significance of lumbosacral transitional anomalies. Acta Orthop Belg. 2007;73(6):687–95.
- Mahato NK. Morphological traits in sacra associated with complete and partial lumbarization of first sacral segment. Spine J. 2010;10(10):910–5.
- Panjabi MM, Goel V, Oxland T, et al. Human lumbar vertebrae: quantitative three-dimensional anatomy. Spine. 1992;17(3).
- Masharawi Y, Salame K, Mirovsky Y, et al. Vertebral body shape variation in the thoracic and lumbar spine: characterization of its asymmetry and wedging. Clin Anat. 2008;21(1):46–54.
- Li B, Jiang B, Fu Z, et al. Accurate determination of isthmus of lumbar pedicle: a morphometric study using reformatted computed tomographic images. Spine (Phila Pa 1976). 2004;29(21):2438–44.
- Su BW, Kim PD, Cha TD, et al. An anatomical study of the mid-lateral pars relative to the pedicle footprint in the lower lumbar spine. Spine (Phila Pa 1976). 2009;34(13):1355–62.
- Robertson PA, Stewart NR. The radiologic anatomy of the lumbar and lumbosacral pedicles. Spine (Phila Pa 1976). 2000;25(6):709–15.
- Chadha M, Balain B, Maini L, et al. Pedicle morphology of the lower thoracic, lumbar, and S1 vertebrae: an Indian perspective. Spine (Phila Pa 1976). 2003;28(8):744–9.
- Zindrick MR, Wiltse LL, Doornik A, et al. Analysis of the morphometric characteristics of the thoracic and lumbar pedicles. Spine. 1987;12(2):160.
- Barber JW, Boden SD, Ganey T, et al. Biomechanical study of lumbar pedicle screws: does convergence affect axial pullout strength? J Spinal Disord. 1998;11(3):215–20.
- Roy-Camille R, Saillant G, Mazel C. Internal fixation of the lumbar spine with pedicle screw plating. Clin Orthop Relat Res. 1986;203:7–17.
- Magerl FP. Stabilization of the lower thoracic and lumbar spine with external skeletal fixation. Clin Orthop Relat Res. 1984;189:125–41.
- Weinstein JN, Spratt KF, Spengler D, et al. Spinal pedicle fixation: reliability and validity of roentgenogram-based assessment and surgical factors on successful screw placement. Spine (Phila Pa 1976). 1988;13(9):1012–8.
- Krenz J, Troup JD. The structure of the pars interarticularis of the lower lumbar vertebrae and its relation to the etiology of spondylolysis, with a report of a healing fracture in the neural arch of a fourth lumbar vertebra. J Bone Joint Surg Br. 1973;55(4):735–41.
- Bogduk N. Clinical anatomy of the lumbar spine and sacrum. UK: Churchill Livingstone; 2005.
- Hadley LA. Fatigue fracture of the fifth lumbar neural arch; is spondylolysis a stress fracture? Clin Orthop. 1955;6:110–3.
- Cavalier R, Herman MJ, Cheung EV, et al. Spondylolysis and spondylolisthesis in children and adolescents: I. Diagnosis, natural history, and nonsurgical management. J Am Acad Orthop Surg. 2006;14(7):417–24.
- Fredrickson BE, Baker D, McHolick WJ, et al. The natural history of spondylolysis and spondylolisthesis. J Bone Joint Surg Am. 1984;66(5):699–707.
- Standring S. Gray's anatomy. UK: Churchill Livingstone; 2008.
- Louyot P. [Comments on the accessory tubercle of the lumbar costiform apophysis (author's transl)]. J Radiol Electrol Med Nucl. 1976;57(12):905.
- Ullrich CG, Binet EF, Sanecki MG, et al. Quantitative assessment of the lumbar spinal canal by computed tomography. Radiology. 1980;134(1):137–43.
- Inufusa A, An HS, Lim T-H, et al. Anatomic changes of the spinal canal and intervertebral foramen associated with flexion-extension movement. Spine. 1996;21(21):2412–20.
- Schonstrom NS, Bolender NF, Spengler DM. The pathomorphology of spinal stenosis as seen on CT scans of the lumbar spine. Spine (Phila Pa 1976). 1985;10(9):806–11.
- Vital JM, Lavignolle B, Grenier N, et al. Anatomy of the lumbar radicular canal. Anat Clin. 1983;5(3):141–51.
- Hasegawa T, An HS, Haughton VM, et al. Lumbar foraminal stenosis: critical heights of the intervertebral discs and foramina. A cryomicrotome study in cadavera. J Bone Joint Surg Am. 1995;77(1):32–8.
- Hasue M, Kikuchi S, Sakuyama Y, et al. Anatomic study of the interrelation between lumbosacral nerve roots and their surrounding tissues. Spine. 1983;8(1):50.
- Vialle R, Levassor N, Rillardon L, et al. Radiographic analysis of the sagittal alignment and balance of the spine in asymptomatic subjects. J Bone Joint Surg Am. 2005;87(2):260–7.
- Heller JG, Amrani J, Hutton WC. Transverse ligament failure: a biomechanical study. J Spinal Disord. 1993;6(2):162–5.
- Dickman CA, Mamourian A, Sonntag VK, et al. Magnetic resonance imaging of the transverse atlantal ligament for the evaluation of atlantoaxial instability. J Neurosurg. 1991;75(2):221–7.
- Dvorak J, Schneider E, Saldinger P, et al. Biomechanics of the craniocervical region: the alar and transverse ligaments. J Orthop Res. 1988;6(3):452–61.
- Tubbs RS, Kelly DR, Humphrey ER, et al. The tectorial membrane: anatomical, biomechanical, and histological analysis. Clin Anat. 2007;20(4):382–6.
- Iatridis JC, MacLean JJ, O'Brien M, et al. Measurements of proteoglycan and water content distribution in human lumbar intervertebral discs. Spine (Phila Pa 1976). 2007;32(14):1493–7.
- Antoniou J, Steffen T, Nelson F, et al. The human lumbar intervertebral disc: evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J Clin Invest. 1996;98(4):996–1003.
- Roberts S, Evans H, Trivedi J, et al. Histology and pathology of the human intervertebral disc. J Bone Joint Surg Am. 2006;88 Suppl 2:10–4.
- Brodin H. Paths of nutrition in articular cartilage and intervertebral discs. Acta Orthop Scand. 1955;24(3):177–83.
- Marchand F, Ahmed AM. Investigation of the laminate structure of lumbar disc anulus fibrosus. Spine (Phila Pa 1976). 1990;15(5):402–10.
- Chelberg MK, Banks GM, Geiger DF, et al. Identification of heterogeneous cell populations in normal human intervertebral disc. J Anat. 1995;186(Pt 1):43–53.
- Nosikova YS, Santerre JP, Grynpas M, et al. Characterization of the annulus fibrosus-vertebral body interface: identification of new structural features. J Anat. 2012;221(6):577–89.
- Smith LJ, Fazzalari NL. Regional variations in the density and arrangement of elastic fibres in the anulus fibrosus of the human lumbar disc. J Anat. 2006;209(3):359–67.
- Yu J, Fairbank JC, Roberts S, et al. The elastic fiber network of the anulus fibrosus of the normal and scoliotic human intervertebral disc. Spine (Phila Pa 1976). 2005;30(16):1815–20.
- Chan WC, Sze KL, Samartzis D, et al. Structure and biology of the intervertebral disk in health and disease. Orthop Clin North Am. 2011;42(4):447–64, vii.
- Jahnke MR, McDevitt CA. Proteoglycans of the human intervertebral disc. Electrophoretic heterogeneity of the aggregating proteoglycans of the nucleus pulposus. Biochem J. 1988;251(2):347–56.
- Nachemson ALF, Morris JM. In vivo measurements of intra-discal pressure discometry, a method for the determination of pressure in the lower lumbar discs. J Bone Joint Surg Am. 1964;46(5):1077–92.
- Roberts S, Menage J, Urban JP. Biochemical and structural properties of the cartilage end-plate and its relation to the intervertebral disc. Spine (Phila Pa 1976). 1989;14(2):166–74.
- Urban JP, Smith S, Fairbank JC. Nutrition of the intervertebral disc. Spine (Phila Pa 1976). 2004;29(23):2700–9.
- Nachemson A. The effect of forward leaning on lumbar intradiscal pressure. Acta Orthop. 1965;35(1-4):314–28.
- Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine (Phila Pa 1976). 1988;13(2):173–8.
- Buckwalter JA. Aging and degeneration of the human intervertebral disc. Spine (Phila Pa 1976). 1995;20(11):1307–14.
- Frobin W, Brinckmann P, Kramer M, et al. Height of lumbar discs measured from radiographs compared with degeneration and height classified from MR images. Eur Radiol. 2001;11(2):263–9.
- Francis CC. Variations in the articular facets of the cervical vertebrae. Anat Rec. 1955;122(4):589–602.
- Lee DJ, Winkelstein BA. The failure response of the human cervical facet capsular ligament during facet joint retraction. J Biomech. 2012; 45(14):2325–9.
- Panjabi MM, White AA, Johnson RM. Cervical spine mechanics as a function of transection of components. J Biomech. 1975;8(5):327–36.
- Varlotta G, Lefkowitz T, Schweitzer M, et al. The lumbar facet joint: a review of current knowledge: part 1: anatomy, biomechanics, and grading. Skeletal Radiol. 2011;40(1):13–23.
- Cyron B, Hutton W. The tensile strength of the capsular ligaments of the apophyseal joints. J Anat. 1981;132(Pt 1):145.
- Giles LGF. Human lumbar zygapophyseal joint inferior recess synovial folds: A light microscope examination. Anat Rec. 1988;220(2):117–24.
- Nachemson A. Lumbar intradiscal pressure. Experimental studies on post-mortem material. Acta Orthop Scand Suppl. 1960;43:1–104.
- Grobler LJ, Robertson PA, Novotny JE, et al. Etiology of spondylolisthesis. Assessment of the role played by lumbar facet joint morphology. Spine (Phila Pa 1976). 1993;18(1):80–91.
- Lorenz M, Patwardhan A, Vanderby R, Jr. Load-bearing characteristics of lumbar facets in normal and surgically altered spinal segments. Spine (Phila Pa 1976). 1983;8(2):122–30.
- Yang KH, King AI. Mechanism of facet load transmission as a hypothesis for low-back pain. Spine (Phila Pa 1976). 1984;9(6):557–65.
- Taylor JR, Twomey LT. Age changes in lumbar zygapophyseal joints. Observations on structure and function. Spine (Phila Pa 1976). 1986;11(7):739–45.
- Van Schaik JP, Verbiest H, Van Schaik FD. The orientation of laminae and facet joints in the lower lumbar spine. Spine (Phila Pa 1976). 1985;10(1):59–63.
- Ahmed AM, Duncan NA, Burke DL. The effect of facet geometry on the axial torque-rotation response of lumbar motion segments. Spine (Phila Pa 1976). 1990;15(5):391–401.
- Nagaosa Y, Kikuchi S, Hasue M, et al. Pathoanatomic mechanisms of degenerative spondylolisthesis: a radiographic study. Spine. 1998; 23(13):1447–51.
- Dai L. Orientation and tropism of lumbar facet joints in degenerative spondylolisthesis. Int Orthop. 2001;25(1):40–2.
- Grobler LJ, Robertson PA, Novotny JE, et al. Etiology of spondylolisthesis. Assessment of the role played by lumbar facet joint morphology. Spine. 1993;18(1):80.
- Fujiwara A, Tamai K, An HS, et al. Orientation and osteoarthritis of the lumbar facet joint. Clin Orthop Relat Res. 2001;385:88–94.
- Masharawi Y, Dar G, Peleg S, et al. Lumbar facet anatomy changes in spondylolysis: a comparative skeletal study. Eur Spine J. 2007;16(7):993–9.
- White AA, Panjabi MM. Clinical Biomechanics of the Spine. Philadelphia: Lippincott-Raven; 1990.
- Francois RJ. Ligament insertions into the human lumbar vertebral body. Acta Anat (Basel). 1975;91(3):467–80.
- Devin Leahy P, Puttlitz CM. The effects of ligamentous injury in the human lower cervical spine. J Biomech. 2012; 45(15):2668–72.
- Tsuyama N. Ossification of the posterior longitudinal ligament of the spine. Clin Orthop Relat Res. 198;(184):71.
- Mercer SR, Bogduk N. Clinical anatomy of ligamentum nuchae. Clin Anat. 2003;16(6):484–93.
- Tanaka N, Fujimoto Y, An HS, et al. The anatomic relation among the nerve roots, intervertebral foramina, and intervertebral discs of the cervical spine. Spine. 2000;25(3):286–91.
- Soleiman J, Demaerel P, Rocher S, et al. Magnetic resonance imaging study of the level of termination of the conus medullaris and the thecal sac: influence of age and gender. Spine (Phila Pa 1976). 2005;30(16):1875–80.
- Needles J. The caudal level of termination of spinal cord in American whites and American negroes. Anat Rec. 1935;63:417–24.
- Nauta HJ, Dolan E, Yasargil MG. Microsurgical anatomy of spinal subarachnoid space. Surg Neurol. 1983;19(5):431–7.
- Wall EJ, Cohen MS, Massie JB, et al. Cauda equina anatomy. I: Intrathecal nerve root organization. Spine (Phila Pa 1976). 1990;15(12):1244–7.
- Bouchard JM, Copty M, Langelier R. Preoperative diagnosis of conjoined roots anomaly with herniated lumbar disks. Surg Neurol. 1978;10(4):229–31.
- Villiger E, Piersol GA. Brain and spinal cord: a manual for the study of the morphology and fibre-tracts of the central nervous system. Philadelphia: J. B. Lippincott & Co.; 1918.
- Cohen MS, Wall EJ, Brown RA, et al. Cauda equina anatomy. II: Extrathecal nerve roots and dorsal root ganglia. Spine (Phila Pa 1976). 1990;15(12):1248–51.
- Foye PM, Buttaci CJ, Stitik TP, et al. Successful injection for coccyx pain. Am J Phys Med Rehabil. 2006;85(9):783–4.
- Heary RF, Albert TJ, Ludwig SC, et al. Surgical anatomy of the vertebral arteries. Spine. 1996;21(18):2074–80.
- Russo VM, Graziano F, Peris-Celda M, et al. The V2 segment of the vertebral artery: anatomical considerations and surgical implications. J Neurosurg Spine. 2011;15(6):610–9.
- Lu J, Ebraheim NA, Georgiadis GM, et al. Anatomic considerations of the vertebral artery: implications for anterior decompression of the cervical spine. J Spinal Disord Tech. 1998;11(3):233–6.
- Neo M, Matsushita M, Iwashita Y, et al. Atlantoaxial transarticular screw fixation for a high-riding vertebral artery. Spine. 2003;28(7):666–70.
- Paramore CG, Dickman CA, Sonntag VKH. The anatomical suitability of the C1-2 complex for transarticular screw fixation. J Neurosurg. 1996;85(2):221–4.
- Madawi AA, Solanki G, Casey ATH, et al. Variation of the groove in the axis vertebra for the vertebral artery implications for instrumentation. J Bone Joint Surg Br. 1997;79(5):820–3.
- Ebraheim NA, Xu R, Ahmad M, et al. The quantitative anatomy of the vertebral artery groove of the atlas and its relation to the posterior atlantoaxial approach. Spine (Phila Pa 1976). 1998;23(3):320–3.
- Currier BL, Maus TP, Eck JC, et al. Relationship of the internal carotid artery to the anterior aspect of the C1 vertebra: implications for C1-C2 transarticular and C1 lateral mass fixation. Spine (Phila Pa 1976). 2008;33(6):635–9.
- Kuklo TR, Lehman RA, Lenke LG. Structures at risk following anterior instrumented spinal fusion for thoracic adolescent idiopathic scoliosis. J Spinal Disord Tech. 2005;18 Suppl:S58-64.
- Hyodoh H, Shirase R, Kawaharada N, et al. MR angiography for detecting the artery of Adamkiewicz and its branching level from the aorta. Magn Reson Med Sci. 2009;8(4):159–64.
- Hassen-Khodja R, Batt M, Michetti C, et al. Radiologic anatomy of the anastomotic systems of the internal iliac artery. Surg Radiol Anat. 1987;9(2):135–40.
- Ratcliffe J. The anatomy of the fourth and fifth lumbar arteries in humans: an arteriographic study in one hundred live subjects. J Anat. 1982;135(Pt 4):753.
- Parsons F, Keith A. Sixth Annual Report of the Committee of Collective Investigation of the Anatomical Society of Great Britain and Ireland, 1895-96. J Anat Physiol. 1896;31(Pt 1):31.
- Poynter CWMC. Congenital Anomalies of the Arteries and Veins of the Human Body: With Bibliography. University of Nebraska; 1922.